|
Means of
transferring heat
Conduction
- The transfer of heat by
either contact between the heat source and the
object to be heated or within the object from one
point to another
- An example would be a
coffee pot on a warming plate
Convection

- The transfer of heat,
from the heat source to the object being heated,
via a fluid medium. That medium is commonly air.
- An example would be a
preheat oven used in front of an infrared tunnel
on a thermoforming machine or a convection oven
used in a paint curing application
Radiation
- The transfer of heat via
electromagnetic radiation between the heat source
and the object to be heated.
- Radiation is broken down
into many subsets divided by different
wavelengths. Some of which are:
- Ultra Violet
- Infrared
- Microwave
- Radio Frequency
The Electromagnetic Spectrum

The Infrared Spectrum
Infrared heating applications

|

|
Thermoforming
|
Powder
Coating
|
Infrared Specifications
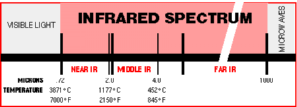
- The wavelength spectrum
ranging from .72 to 1000 microns.
- The infrared region is
divided into 3 subsets
- Short wave (near) .72 - 2 microns
(7000-2150°F)
- Medium wave (middle) 2 - 4 microns
(2150-845°F)
- Long wave (far) 4 - 1000 microns
(845-<32°F)
- The useful infrared region for
industrial process heating ranges from 1.17 to 5.4 microns (4000°F
- 500°F).
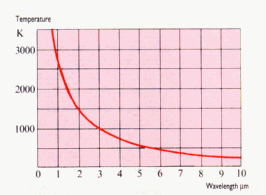
- The wavelength is
inversely proportional to temperature. As the
temperature goes up, the wavelength goes down.
The history of infrared heating
- First used back in the
1930's for automotive paint curing applications
- It wasn't until World War
II that infrared heating came into heavy use. It
speed up the production of military equipment.
- After WW II the use of
infrared heating once again slowed down.
- Today, the use of
infrared heating is growing rapidly around the
world. Utility Technology Centers have helped to
spawn that growth.
Why use infrared heating systems?
- Reduces floor space
- Lowers energy consumption
- Increases line speed
- Reduces maintenance
- Clean operating
environment
 Daily examples of
infrared heating Daily examples of
infrared heating
- Toaster
- Bathroom heat lamp
- Barbecue grill
- Light bulb (90% heat -
10% light)
- The granddaddy of all
infrared heaters - The Sun. Half of the sun's
energy is infrared radiation
What is infrared heating?
- The electromagnetic energy that is
emitted by all bodies above -273°C (0°K or absolute zero).
- When infrared energy
strikes an object it causes the surface electrons
to excite and oscillate.
- This oscillation creates
heat.
- It travels in straight
lines from the source
- It can be directed into
specific patterns with the use of properly
designed reflectors
- It decreases in intensity
as it travels outward from its source
How does heater output wavelength
effect the process?
- Infrared radiation is
either
- Reflected
- Absorbed
- Transmitted (meaning to
pass through)
- Materials have different
absorption curves
- Ideally, you would like
the heater to output the majority of its energy
in the area where it is best absorbed.
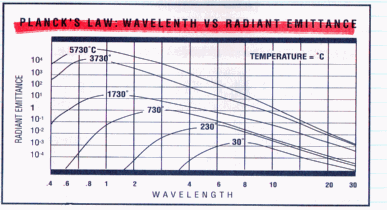
Plank's Law
- Plank's Law defines the
relationship of wavelength output to temperature
based on a point source in vacuum. Raising
the output power increased the temperature of the
point source. This resulted in the peak
wavelength shifting to a shorter wavelength, as
displayed in the above curve.
Stefan Boltzmann Law
- F = s
T4 s
= 5.73 x 10-8 W/m 2 x
K 4
- The total energy radiated
is equal to the black body temperature to the
fourth power
- That is to say - if the
temperature of an infrared heater is doubled,
then the power output will increase by sixteen
fold
- The peak wavelength will
shift to a shorter wavelength
Wien's Law
- This curve and formula
express the relationship between wavelength and
absolute temperature
The Inverse Square Law
- This law is applicable to
a point source, not necessarily a real
life infrared emitter
- The radiant intensity at
the product to be heated varies inversely as the
square of it's distance from the emitter surface
- In real life applications
the law does not hold true. View factor is a
better determinate of the radiant loss due to
distance from the product to the heater
View Factor
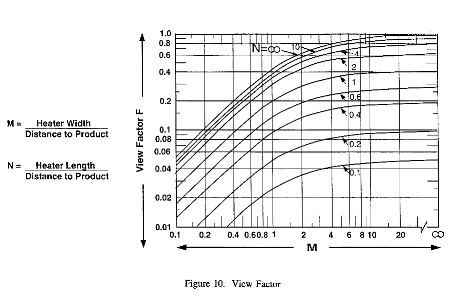
Compliments of CMF - Center for
Materials Fabrication
Heating Technologies for Thermoforming
Definitions
Emissivity the
relationship between reflectivity and absorption. A
perfect absorber (black body) has an emissivity of
1.0. The perfect reflector has an emissivity of 0.
All products fall somewhere in between this range.
Color Sensitivity refers to different curing or
heating rates based on the emitter wavelength. White
coatings are more reflective and therefore do not
absorb as much infrared energy. Therefore, white
coatings take much longer to heat up. This factor is
more acute with shorter wavelengths.
An
example of color sensitivity
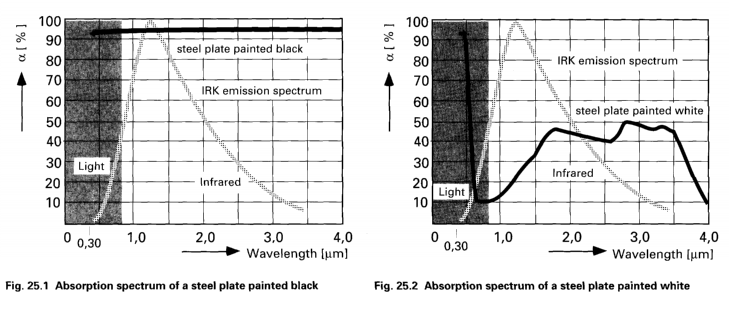
An
example of color sensitivity
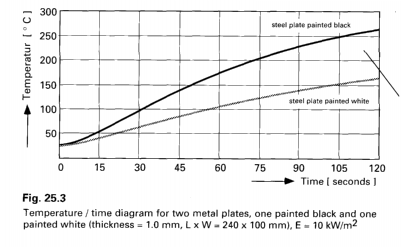
A quick comparison between
emitters at different wavelengths
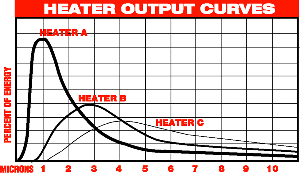
Wavelength discussion
- The peaks
for most plastics are at 3.5 and 6-10 microns
- 3.5 microns equates to
approximately 1030° F
- 6 microns equates to a
temperature below 500° F
- In order
to reduce the heating cycle time, the heater
output is set at the highest possible
temperature, without burning the sheet.
- The goal
is to put in as much heat as possible, without
damaging the product surface - at any wavelength
- Wavelength,
radiant efficiency, and power output all
determine how quickly the sheet can be heated. It
also determines how much energy is required.
Absorption curves
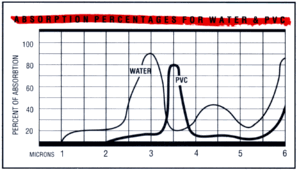
Absorption curves
Courtesy
of EPRI/CMF
Technology Guidebook for Electric Infrared
Process Heating |
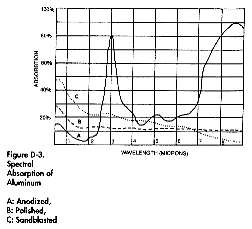 |
Typical drying and
hardening curves
Courtesy
of EPRI/CMF
Technology Guidebook
for
Electric Infrared Process Heating
- A
standard convection oven transfers
500-2,000 BTU/hour - square foot while IR
ovens transfer from 3000 - 25,000
BTU/hour - square foot
|
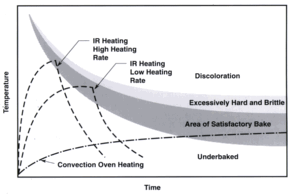 |
IR vs Convection Heat
Transfer Comparison
- This
curve was found in Electric Process
Heating By Maurice
Orfeuil, Battelle Press
|
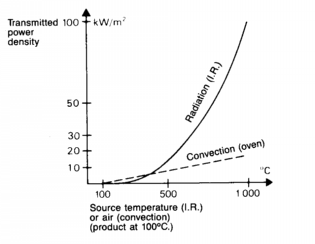 |
Chart comparing IR
to Convection Heating
Courtesy
of EPRI/CMF Technology Guidebook for Electric
Infrared Process Heating
0.05" Steel & Aluminum
|
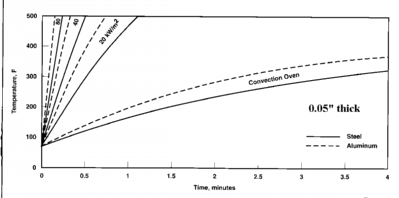 |
Chart comparing IR
to Convection Heating
Courtesy
of EPRI/CMF
Technology Guidebook for Electric Infrared
Process Heating
0.25" Steel & Aluminum
|
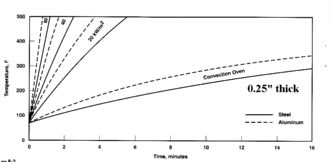 |
Chart comparing IR to
Convection Heating
Courtesy
of EPRI/CMF
Technology Guidebook for Electric Infrared
Process Heating
1.0" Steel & Aluminum
|
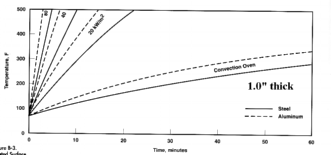 |
Chart comparing
IR to Convection Heating
Courtesy
of EPRI/CMF
Technology Guidebook for Electric Infrared
Process Heating
0.25" Plastic & Wood
|
 |
Typical Infrared Misconceptions
Misconception
#1
Courtesy of EPRI/CMF
Technology Guidebook for Electric Infrared Process
Heating
- IR
Radiation may be harmful to oven operators.
- There is
no immediate danger associated with the use of IR
radiation compared with ultraviolet radiation or
microwave. However, as a precaution one should
avoid prolonged viewing of high intensity IR
emitters at close distances (less than 15 feet).
Repeated, long term, near exposure to high
intensity IR radiation may cause cataracts in
some individuals.
Misconception
#2
Courtesy of EPRI/CMF
Technology Guidebook for Electric Infrared Process
Heating
- IR ovens
are effective in heating only flat surfaces.
- Flat
surfaces are ideally suited to heating by IR
radiation. They can be heated rapidly and
effectively in an IR oven. However, more complex,
three-dimensional shapes can also be heated in an
IR oven. Three-dimensional parts can be rotated
so that all sides are evenly exposed to radiation
as they pass through the oven. The heating rate
can also be varied from zone to zone to allow
sufficient soak times to heat internal regions of
a part.
Misconception
#3
Courtesy of
EPRI/CMF
Technology Guidebook for Electric Infrared Process
Heating
- IR
radiation works better in a vacuum with little or
no air moving.
- Air is
virtually transparent to IR radiation. IR
radiation is neither absorbed nor scattered by
air. However, water vapor, carbon dioxide, and
other greenhouse gases do absorb IR radiation. If
the air between the emitter and the product
contains water vapor or other absorbing gases, it
could absorb a portion of the IR radiation. For
distances between the emitter and the absorber of
a few feet or less, the energy absorbed by the
gas will be negligible.
Misconception
#4
Courtesy of EPRI/CMF
Technology Guidebook for Electric Infrared Process
Heating
- Short-wavelength
IR penetrates more than medium and
long-wavelength IR.
- Although
this statement is true in many cases, it is not
universally true. For example, metals do not
transmit IR radiation of any wavelength. All the
IR radiation incident on a metal is absorbed or
reflected at the surface. On the other hand, some
non-metals transmit radiation. These include
water, glass, quartz, and some ceramic and
polymer materials. These same materials also may
transmit longer wavelengths to some degree.
Misconception
#5
- Only one
wavelength is best for a given application.
- This
statement is blatantly false. There are many
factors that need to be considered. All
wavelengths will most likely work for a given
application. But you need to consider not only
the heating rate, but also the available floor
space, maintenance requirements, heater
durability, response time, heater and system
efficiency, initial oven cost, energy consumption
cost, conveyor speed, part size variation,
controllability, and aggravation cost. All of
these items need to be considered in order to
pick the right solution.
Bibliography
- Maurice
Orfeuil, Electric Process
Heating, Battelle Press 1987
- JR
O'Connell, EFB Croft, WC Hankins, Electric
Infra-red Heating for Industrial Processes, EA
Technology 1990?
- Technology
Guidebook for Electric Infrared Process Heating,
CMF Report No. 93-2 1993
- Jay
Siedenburg, Heating Technologies for
Thermoforming, CMF Report No. 95-1 1995
- Shelby F
Thames, Ph.D., Presentation on the Use of IR
with Polymer Applications, IREA meeting 1997
- Thomas A.
Stryker, The Heat Processing Handbook for
Paint & Powder Applications, 1997
- Philips
Lighting Application Information, 1994
|

 Daily examples of
infrared heating
Daily examples of
infrared heating